Teflon (PTFE) because of its excellent chemical stability of high temperature resistance, known as the “plastic king”, it is often used as a membrane separation material. But because the material molecular structure is highly symmetrical, causing its surface hydrophobicity to be strong, this affects Teflon in the bonding and liquid filtration and other applications. This question had been troubled Teflon coated fabric suppliers for many years. In order to make up for these shortcomings, the major Teflon coated suppliers in the world spent a huge investment on the PTFE membrane modified.
At present, the major suppliers commonly used chemical method, high temperature method and plasma method and other methods to deal with PTFE membrane surface. But the surface of the Teflon film treated by the chemical method is obviously darkened and the ontology of the material is affected. PTFE will evaporate toxic substances.in high temperature melting process. And the plasma method has the effect of changing the surface properties of the material in a short time without affecting the inherent properties of the substrate, and the process is dry processing, saving energy protection environment.
In this paper, Ar is used as the treatment medium to treat the Teflon membrane, the grafted AA monomer further enhances its hydrophilicity to address the timeliness of plasma treatment. The effects of different plasma treatments and grafting processes on the hydrophilicity of Teflon films were investigated, and the contact angle was used to characterize the hydrophilicity of FTIR. The solution and the corresponding results in this paper, there have a huge reference value for the suppliers of Teflon coated fabric, Teflon adhesive tape and Teflon conveyor belt.
Experiment
Major reagents and equipment
Teflon hollow fiber membrane, Lab homemade; Argon (purity ≥99.99%), Acrylic acid, anhydrous ethanol (AR).
DSA25S contact angle measuring instrument, Germany Kruss company; BTF-1200C-S-SL plasma processing system; IR Affinity-1 Fourier transform infrared spectrometer.
Sample preparation
Ar plasma pretreatment
The Teflon membrane was ultrasonically cleaned with absolute ethanol for 30 min, after drying, put it in the reaction chamber of the quartz stent, Vacuum to 2 ~ 6Pa, through argon, draining the residual air in the reaction chamber, adjust the needle to control the gas flow in 10~60cm3·min-1, adjust the pressure to 20 ~ 100Pa, After stabilization, start the RF power source, the Teflon film was pretreated by plasma, processing power of 100 ~ 500W, processing time of 30 ~ 300s.
Surface graft polymerization
After the plasma pretreatment of the sample with air contact oxidation for a period of time, put in the concentration of 5% to 45% of the AA solution, heated to 3070, the processing time is 214h, After the reaction, remove it and wash it in 40 distilled water for 12 hours.
Performance test and structural characterization
Contact angle
The contact angle of the PTFE film surface was measured using a contact angle meter, and the measurement was carried out at a room temperature and humidity of 30% RH, using micro-injectors to control the droplets in about 2μL, the average of the five points measured on the same sample surface is taken as the final contact angle.
ATR-FTIR
The change of membrane surface structure before and after modification was analyzed by attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR), ATR-FTIR analysis of the scanning frequency is 30 times, scanning range is 600 ~ 4500cm-1, the minimum resolution is 2cm-1.
Results and discussion
Effect of Plasma Treatment Conditions on Surface Wettability of Teflon Membrane
By testing the plasma processing power time and the gas flow rate of PTFE membrane surface contact angle, explore the best conditions for plasma treatment.
Effect of Plasma Treatment Power on Surface Wettability of PTFE Membrane
The effect of the contact angle of the film surface on the treated power is shown in Table 1, Membrane surface wettability with power changes shown in Figure 1.
Table1 Effect of power in Ar plasma on PTFE membrane contact angle
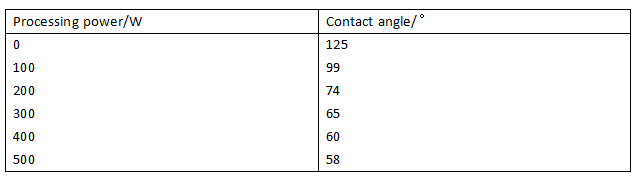

Fig.1 Teflon membrane wettability change with different plasma powers
As can be seen from Table 1, The contact angle of the surface of the Teflon membrane is 125°. When the power increases from 0W to 300W, the contact angle decreases from 125°to 65°. When the power increases from 300W to 500W, the contact angle becomes 58°.As the power increases, the contact angle of the membrane surface initially decreases rapidly and then slowly and stabilizes. Figure 1 shows that the wettability of the membrane with the power increase gradually improved, and ultimately no longer significantly changed.
The reason is that the level of power represents the amount of energy that can be accepted on the surface of the membrane, and the power increases so that the energy obtained by the quantitative argon molecules also increases, the chemical bond of the membrane surface molecules is increased by the probability of being opened, and the number of hydrophilic groups formed by further combining the free radicals in the plasma increases, thereby reducing the contact angle of the film surface. However, when the power is too large, the plasma generated free radicals between the probability of binding will increase, can not effectively combine the free surface of the membrane surface.
Considering, the best processing power is 300W.
Effect of Plasma Treatment Time on Surface Wettability of Teflon Membrane
By changing the treatment time, to study its effect on the film surface wettability, the results shown in Figure 2
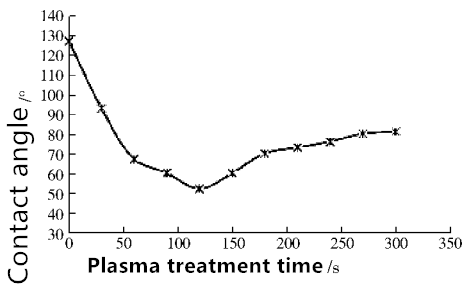
Fig. 2 Teflon membrane contact angle change with different plasma times
It can be seen from Fig. 2 that the contact angle of the membrane surface is different when the processing time is different, and the contact angle increases gradually with time, and then begins to rise and stabilize after a minimum value. The contact angle decreases rapidly in a short time and reaches a minimum of 52°at 120 s, and the contact angle increases after more than 120 s.
The reason may be related to the mechanism of plasma reaction. As shown in Figure 3. When the time is short, the active particles in the Ar plasma bombard the membrane surface, resulting in the membrane surface of the C-F bond fracture, generating peroxides, These free radicals formed by the destruction of the membrane matrix recombine with other free radicals in the plasma, forming a polar group on the membrane surface, thereby significantly improving the wettability of the film surface. When the time reaches a certain limit, the free radicals generated on the membrane surface also reach a critical value, and then extended the time will increase the probability of the new generation of free radicals between the cross-linking reaction until the membrane surface polarity The rate of increase to achieve dynamic equilibrium, contact angle is no longer significantly increased.
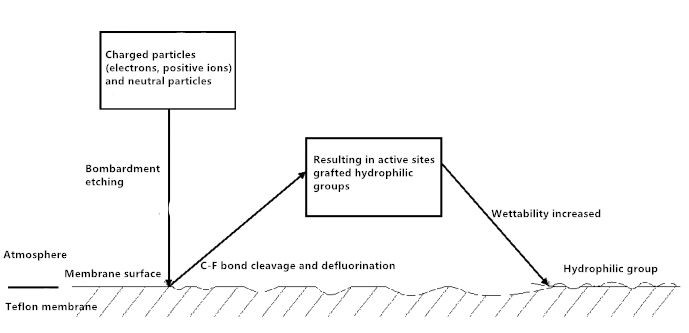
Fig. 3 The principle of plasma treatment on the hydrophilic modification of PTFE membrane
According to the above, both to ensure that the hydrophilic modification effect can improve the experimental efficiency of the best processing time is 120s.
Effect of Plasma Flow Rate on Surface Wettability of PTFE Membrane
Table 2 Effect of different plasma gas flows on PTFE membrane contact angle
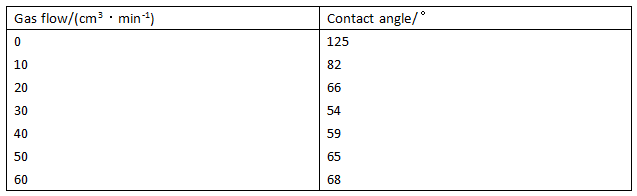
The effect of the gas flow on the film surface wettability was investigated by changing the gas flow rate. The results are shown in Table 2.
It can be seen from Table 2 that when the gas flow rate is 30cm3·min-1, the contact angle of the membrane surface is at least 54°, which is 61° lower than that of the original film. When the flow rate is increased to 50cm3·min- The contact angle becomes 65°.
As the gas flow increases, the contact angle of the membrane surface decreases first. This is because as the gas flow increases, the concentration of argon ions in the plasma generator increases rapidly, but also accelerates the rate of regeneration of the ionized gas, Resulting in enhanced deflagration on the membrane surface, thus generating more free radicals, a large amount of hydrophilic groups are polymerized on the surface of the film so that the contact angle after treatment decreases first. But when the gas flow exceeds 40cm3·min-1, the contact angle of the membrane surface increases. This is due to the increase in gas flow, resulting in reduced vacuum in the plasma generator, shortening the activity of high-energy particles free path, and thus can not fully stimulate the gas molecules in the reactor.
Effect of Grafting Conditions on Surface Wettability of Teflon Membrane
Effect of Acrylic Acid Concentration on Surface Wettability of Teflon Membrane
The concentration of AA monomer has a great effect on the wettability of the membrane surface, as shown in Fig 4.
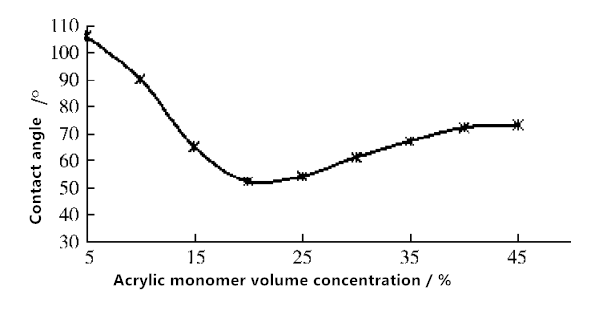
Fig. 4 PTFE membrane contact angle change with different AA volume concentrations
It can be seen from Fig. 4 that the wettability of the membrane surface increases with the increase of AA concentration, and the contact angle decreases to 52° when the AA concentration reaches 20%. This is because when the AA concentration is low, the solution viscosity in the reactor is low, the monomer is easy to approach the free radicals on the membrane surface, which can lead to the graft polymerization. When the AA concentration continues to increase, the contact angle is slightly increased. This is because when the AA concentration is too high, the AA monomer self-aggregation or homopolymerization rate is accelerated, so that the monomer can not react with the free radicals on the membrane surface.
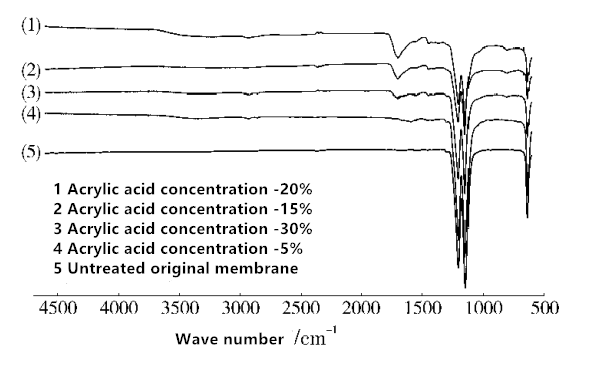
Fig. 5 Infrared spectrogram of Teflon membrane with different AA volume concentrations
The ATR-FTIR test was performed on the membrane surface. The results are shown in Fig.5. It can be seen from Fig. 5 that the plasma treated membranes have peaks that characterize AA grafts, and the positions and shapes of the peaks are basically the same. When the AA concentration of 20%, the measured bands appear rich in bands near 3300 cm-1, should be -OH stretching vibration peak, the band at 2800-3000 cm-1 is the stretching vibration of saturated C-H, near 1730cm-1 appear the C = O stretching vibration peak, 1149cm-1 and 1210cm-1 are C-F stretching vibration peaks. These characteristic peaks demonstrate that AA is successfully grafted onto the membrane surface.
Effect of Grafting Reaction Time and Temperature on Surface Wettability of PTFE Membrane
The effect of graft reaction time and temperature on the contact angle of the membrane surface is shown in Fig.6.
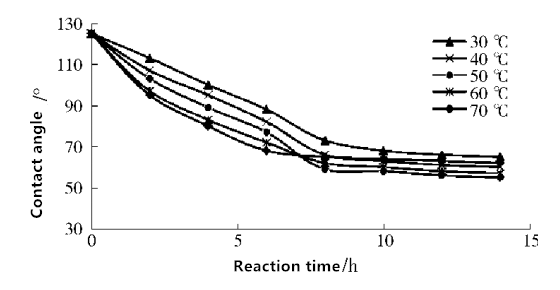
Fig. 6 Effect of different grafting times and temperatures on PTFE membrane wettability
As can be seen from Figure 6, As the temperature increases, the contact angle of the membrane surface gradually decreases, and when the temperature is 50 ℃, the minimum contact angle is 58°. This is because the temperature rise makes the free radicals on the membrane surface and the AA monomer in the solution activated, the contact angle in the early reaction with the temperature rise and decreased. When the temperature is more than 50 ℃, the contact angle be increased. May be caused by high temperature self-polymerization of AA monomer, so that the solution becomes viscous and hinder the grafting rate.
Conclusion
The results of this paper show that, Teflon hollow fiber membrane by Ar plasma treatment and then grafted AA monomer, the number of hydrophilic groups on the membrane surface increased significantly, surface energy and surface activity increased, the hydrophilicity is greatly improved and the modification effect is long, Thus broadening the scope of application of Teflon hollow fiber membrane for membrane separation systems in the field of waste water treatment.
If you need more information about our products, please contact us: infocorefrp@gmail.com ,our engineers will answer you and provide free samples.
![]() Hydrophilic Modification of Teflon Hollow Fiber Membrane by Ar Plasma.pdf
Hydrophilic Modification of Teflon Hollow Fiber Membrane by Ar Plasma.pdf